We decided to learn about batteries and what better way to do that than creating our very own pickle battery?

What you need:
A jar of of pickles
Copper wire
Zinc nails
a light
an electrometer
wire cutters
and last but not least, a tray
A jar of of pickles
Copper wire
Zinc nails
a light
an electrometer
wire cutters
and last but not least, a tray
We took the pickles out of the jar and lined them up on the tray. Then we attached them with the copper wire and the zinc nails. Each pickle had one zinc nail and one copper wire in it. The copper connects to a nail in the pickle next to it. This creates a circuit.
Next, we took the electrometer and tested our battery to see how much electricity it was creating. The most we could get was 2.35 output, which is not enough to light our bulb. What we needed was more pickles (or did we? See what we learned at the bottom).
After adding four more pickles and connecting them to the circuit we already had, we got a 4.2 reading on our electrometer. Would this be enough to light a bulb?
It was enough to light our Christmas light bulb! Amazing!
So, how does it work? Surely the pickles aren't full of electricity, are they?
So, how does it work? Surely the pickles aren't full of electricity, are they?
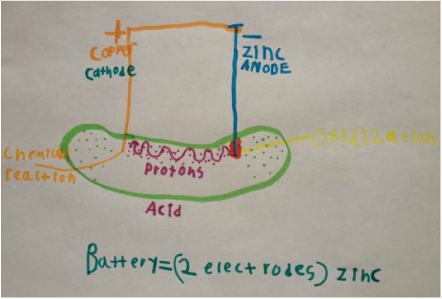
What you need is two electrodes and one electrolyte. The electrodes are the metal: zinc and copper. The electrolyte is the pickle juice. One electrode is willing to give away its electrons: zinc. This makes it have low electronegativity and is called an anode. The copper likes to attract electrons, making it have higher electronegativity than zinc and is called a cathode.
When the zinc is placed in the pickle, a chemical reaction takes place immediately called oxidization (when it happens in places we don't want, we like to call it corrosion). This is where the zinc "gives away" protons and electrons. The electrons travel through the metal, but the protons are too big to do that, so they must travel through the electrolytes: the pickle juice. Once the protons reach the copper, the copper gives the protons an electron, creating another chemical reaction: hydrogen gas.
The electricity comes from the chemical reaction that results from the zinc in the pickle juice and the copper absorbing the protons.
The power actually lies in the zinc, not the pickle! Watch the SciShow video below for an excellent explanation. (We had to watch it a few times to wrap our heads around it!)
When the zinc is placed in the pickle, a chemical reaction takes place immediately called oxidization (when it happens in places we don't want, we like to call it corrosion). This is where the zinc "gives away" protons and electrons. The electrons travel through the metal, but the protons are too big to do that, so they must travel through the electrolytes: the pickle juice. Once the protons reach the copper, the copper gives the protons an electron, creating another chemical reaction: hydrogen gas.
The electricity comes from the chemical reaction that results from the zinc in the pickle juice and the copper absorbing the protons.
The power actually lies in the zinc, not the pickle! Watch the SciShow video below for an excellent explanation. (We had to watch it a few times to wrap our heads around it!)
As stated in the SciShow video, we could've created a lot more electricity by simply connecting more zinc and copper in each pickle, rather than adding more pickles! Who knew? We guess once you understand batteries, it makes perfect sense!



 RSS Feed
RSS Feed
